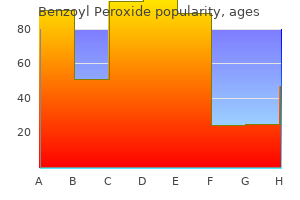
|
STUDENT DIGITAL NEWSLETTER ALAGAPPA INSTITUTIONS |

|
John Joseph Anderson, DPM, FACFAS
Administration of thiazide diuretics reduces urinary calcium excretion and can sometimes cause hypercalcemia acne 5 days after ovulation safe benzoyl 20 gr. The mechanism likely involves an increase in bulk calcium reabsorption with sodium and water in the proximal tubule acne vacuum order benzoyl 20gr without a prescription. Thiazide-induced volume depletion might be the stimulus that triggers this process retinol 05 acne purchase 20 gr benzoyl overnight delivery. Patients receiving systemic cetuximab can develop profound renal magnesium wasting because of the Clin J Am Soc Nephrol 9: 21472163 acne webmd cheap benzoyl 20gr, December, 2014 Distal Convoluted Tubule, Subramanya et al. Chabardes D, Gagnan-Brunette M, Imbert-Teboul M, ґ Gontcharevskaia O, Montegut M, Clique A, Morel F: Adenylate cyclase responsiveness to hormones in various portions of the human nephron. Am J Physiol Renal Physiol 287: F593F601, 2004 Kaissling B, Kriz W: Structural analysis of the rabbit kidney. Adv Anat Embryol Cell Biol 56: 1123, 1979 Dшrup J: Ultrastructure of distal nephron cells in rat renal cortex. J Ultrastruct Res 92: 101118, 1985 Hierholzer K, Wiederholt M: Some aspects of distal tubular solute and water transport. Am J Physiol 250: F1013 F1023, 1986 Yoshitomi K, Shimizu T, Taniguchi J, Imai M: Electrophysiological characterization of rabbit distal convoluted tubule cell. Lourdel S, Paulais M, Cluzeaud F, Bens M, Tanemoto M, Kurachi Y, Vandewalle A, Teulon J: An inward rectifier K(1) channel at the basolateral membrane of the mouse distal convoluted tubule: Similarities with Kir4-Kir5. Am J Physiol Renal Physiol 300: F1385F1393, 2011 Giebisch G: Renal potassium transport: Mechanisms and regulation. Am J Physiol Renal Physiol 285: F289F294, 2003 2162 Clinical Journal of the American Society of Nephrology 74. Lu Z, MacKinnon R: Electrostatic tuning of Mg21 affinity in an inward-rectifier K1 channel. Renal Physiology Collecting Duct Principal Cell Transport Processes and Their Regulation David Pearce,* Rama Soundararajan, Christiane Trimpert, Ossama B. Kohan Abstract the principal cell of the kidney collecting duct is one of the most highly regulated epithelial cell types in vertebrates. The effects of hormonal, autocrine, and paracrine factors to regulate principal cell transport processes are central to the maintenance of fluid and electrolyte balance in the face of wide variations in food and water intake. In marked contrast with the epithelial cells lining the proximal tubule, the collecting duct is electrically tight, and ion and osmotic gradients can be very high. In addition to these essential hormones, additional neuronal, physical, and chemical factors influence Na1, K1, and water homeostasis. Considerable recent progress has improved our understanding of the transporters, receptors, second messengers, and signaling events that mediate principal cell responses to changing environments in health and disease. This review primarily addresses the structure and function of the key transporters and the complex interplay of regulatory factors that modulate principal cell ion and water transport. Ion and water transport in this part of the nephron are highly regulated by a wide variety of stimuli, including hormones, autocrine and paracrine factors, osmotic conditions, and physical factors. In humans, by the time tubular fluid reaches the aldosterone-sensitive distal nephron under physiologic conditions, virtually all amino acids, glucose, bicarbonate, and other nonwaste organic solutes have been removed and water volume has been reduced to approximately 10% of that of glomerular filtrate. Thus, the absolute level of transport of critical ions and water in this nephron segment is markedly lower than in most upstream segments; however, the variability of transport rates is markedly higher. Ion and osmotic gradients between the tubule lumen and the interstitium are also more variable, and are frequently much higher than in other regions. A future article in this series will address the integrated tubule physiology and compare characteristics of different segments. In addition to these essential hormones, other hormones, autacoids, and mechanical factors influence Na1, K1, and water homeostasis. This review primarily addresses the regulation of Na1, K1, and water transport in principal cells. Aldosterone is the primary hormonal regulator of both Na1 and K1 transport, as addressed further below. Cl2 transport is not shown in Figure 1 because it is not a simple function of principal cells; rather, Cl2 transport is a function of both principal and intercalated cells, as well as the paracellular pathway (2,3). This review discusses the key Na1 and K1 pathways operative in principal cells and their regulation, and also describes Cl2 transport. Control of Principal Cell Ion Transport Copyright © 2015 by the American Society of Nephrology 135 136 Clinical Journal of the American Society of Nephrology Figure 1. Like all channels, it does not directly couple Na1 transport to the movement of any other ion or solute. Selectivity is mediated by a signature Gly/Ser-X-Ser sequence, which is adjacent to the amiloride binding site (Figure 2D). These regulatory signals are integrated to produce appropriate levels of Na1 transport to meet physiologic demands. Under physiologic conditions, this effect appears to be mediated by furin and a secondary membrane-resident protease. Furin is a proprotein convertase that resides primarily in the trans-Golgi network and processes proteins transiting through the biosynthetic pathway. It is unclear at this time whether the furin-mediated cleavage is an important locus of regulation or is a device to keep the channel from being turned on prematurely (see below). Plasmin is not present in the tubule lumen under normal conditions; however, in the setting of proteinuria (as seen in the nephrotic syndrome), plasminogen is filtered by the glomerulus and can be converted to plasmin by urokinase, which is present within the tubular lumen (13). The first identified Liddle mutation resulted in a premature translation stop in the b subunit (16), leaving the Na1 pore intact but deleting intracellular target sites for inhibitory control mechanisms (16). Other mutations that cause variable degrees of hyperactivation of the channel were also identified. The key ones required for regulating Na1 transport encode either transporters themselves, or regulatory proteins that control transporter abundance or activity. In fact, most of the aldosterone-induced increase in Na1 transport occurs within the first 3 hours and is primarily mediated by rapidly stimulated genes that encode regulatory proteins, not by increases in transporter gene expression per se. The physiologic role for insulin in the control of Na1 transport has remained obscure; however, its pathophysiologic effects are clear. Kidney tubule Na1 transport remains insulin sensitive, even as other tissues and processes become resistant (37). As insulin levels rise to maintain normal glucose concentration, the retained sensitivity of Na1 transport results in excessive Na1 reabsorption in insulin-resistant states. Further contributing to this problem, the vasodilatory effects of insulin are blunted in insulin-resistant states, and hence only the prohypertensive effects remain (40). This suggestion harkens back to the clinical maxim that "the body defends volume above all else. In particular, autocrine (acting on the same cell) Clin J Am Soc Nephrol 10: 135146, January, 2015 Collecting Duct Principal Cell Transport and Regulation, Pearce et al. The findings of close apposition of sympathetic nerve extensions and principal cells (43), as well as collecting duct expression of b- and a-adrenergic receptors, suggests that principal cell function can be modulated by catecholamines released by efferent renal sympathetic nerves. The nature of such regulation is unclear because differing results have been obtained depending upon the species studied and the agonist utilized. Activation of the peroxisome proliferatoractivated receptor-g by thiazolidinediones is well known to cause Na1 retention. Mice with principal cellspecific knockout of peroxisome proliferator activated receptor-g are resistant to thiazolidinedione-induced Na1 retention (65,66). Salt loading increases renal interstitial adenosine; relatively large amounts of adenosine are found in the inner medulla [reviewed by Rieg and Vallon (68)]. Renin is synthesized and secreted into the tubule lumen by collecting duct cells [reviewed by Navar et al. Luminal renin may bind to prorenin receptors on intercalated cells, enhancing its catalytic activity and modulating intercalated cell function. As addressed below, the bulk Clin J Am Soc Nephrol 10: 135146, January, 2015 Collecting Duct Principal Cell Transport and Regulation, Pearce et al. This is an important detail for clinicians to be aware of, because it is relevant to commonalities among hypertensive diseases, such as Liddle syndrome, apparent mineralocorticoid excess, and primary aldosteronism, as well as to the treatment of saltsensitive essential hypertension. Overstimulation of water retention is found in the syndrome of inappropriate release of the antidiuretic hormone and in activating mutations of the V2R, which both lead to hyponatremia with normovolemia. This involves short-term modulation through alterations in trafficking and longterm regulation through changes in protein expression. First, in principal cells, Cl2 can be reabsorbed or secreted (depending on electrochemical gradient) via the cystic fibrosis transmembrane regulator.
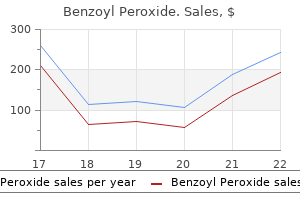
However skin care 5-8 years purchase benzoyl 20 gr without prescription, this connection may only be a recent human adaptation since early hominids may have possessed dark acne red marks order benzoyl 20gr on-line, dense skin care must haves purchase 20 gr benzoyl, terminal body hair acne after stopping birth control generic benzoyl 20 gr without prescription. A closely related primate, the chimpanzee, similar to most other nonhuman primates, exhibits white or lightly pigmented epidermis (591). The tendency toward relative hairlessness in modern humans has been explained by the need to maintain thermal balance under the progressive increase in demands for heat dissipation that results from the enhanced blood flow to the brain. Alternately or complementarily, hairlessness would also reduce parasitic infestations (549). Additional properties of melanin may include a bactericidal potential via the production of orthoquinones (618), and contribution to the tensile strength of hair via cross-linking with proteins. Hair color may have undergone a far more complex evolution than skin pigmentation. Although most humans are dark-haired and dark-eyed, melanization in skin, hair, and eyes do not closely correlate. Indeed, a large fraction of humans have dark eyes and hair but their skin would rate as "white," whereas in some western European populations, black hair commonly coexists with blue eyes. The occurrence of black scalp hair, a potent trap for radiant heat, may appear as a paradoxical development for primates and humans living in tropical climates; however, black scalp hair may provide some protection from sunstroke by helping with the salt balance through the highly efficient and fast ion exchange property by melanin (746, 902). In fact, the pigmented hair on the human scalp may have resulted from the littoral residence of Homo sapiens residing on sea coasts or riverbanks, with diet dominant in fish (many of which concentrate heavy metals). In this context, the capability to rapidly excrete toxic metals provided by the very high turnover of melanized cortical keratinocytes in the pigmented hair shaft would confer a selection advantage (46). Thus the long, melanized scalp hair with its capability to trap and/or bind chemicals, toxins, and heavy metals would prevent their access to living tissues. Pigmented hair may also provide antioxidant defense for the skin and hair follicles due to the high capacity of melanin for binding transition metals. This buffering capacity as applied to calcium would imply a role for melanin in cell function, since calcium is a critical second messenger in pigmentation signaling, acting in the transfer of melanosome to keratinocytes, and in epithelial cell differentiation (746). The epidermis of adult human females is less melanized than in adult males, suggesting a gender-specific effect (626). One possible explanation for this discrepancy could be the higher need for vitamin D in women that is imposed by the Taken together, these data imply that melanin is important for skin homeostasis and that tanning itself represents a distress signal. The same pathophysiological explanation would apply to the localized pigmentation that follows the exposure of melanin to toxic compounds, and that may result in marked increases in melanin granules and melanin deposition (427). Abnormalities in the transfer of melanosomes out of the melanocytes and into receiving keratinocytes represent the human counterpart of the dilute mutation in mice where the motor protein myosin V is defective. Disorders associated with aberrant melanosomes include the macromelanosomes and autophagic giant melanosome complexes of nevocellular nevi, lentigo simplex, malignant melanoma, and the neuroectodermal melanolysomal diseases that include the Elejalde, Chediak, Higashi, and Griscelli syndromes (515, 779, 780). These syndromes are more likely due to disordered melanosome biogenesis than alterations in melanosome degradation. The most common pigment disorders are not disorders of melanin quality, but rather of the pigment-producing cell itself, which may be reduced in number, absent, or hyperactive and commonly, with regional localization. Pigment excess (hypermelanosis) can be associated with inflammatory responses, as in keloid scars, or with local abnormal melanocyte function, as in dysplastic nevi or malignant melanoma. Intact mature melanosomes pass from basal melanocytes into keratinocytes and their lysosomal compartment to become melanin dust in the upper nonviable layers of the epidermis. There is scant information on the actual mechanism of melanin breakdown or biodegradation. Nevertheless, hair melanin granules, unlike those in overlying epidermis, tend to remain intact in the hair shaft. This is especially true for the eumelanogenic melanosomes of the black hair shaft such as seen in the hair of East-Asian (Oriental) individuals, the ethnic group with the highest density of pigment granules. In contrast, the pheomelanin granules characteristic of red and blonde hair, are partially digested. Furry mammals (rodents) Most of the information on skin pigmentation has emerged from the intensive study of rodent coat color, as opposed to research focused specifically on epidermal pigmentation. As a uniquely mammalian trait, hair serves important functions most easily appreciated in furred mammals. These include thermal insulation, camouflage (for many species, melanin affords significant additional protective value. Many furred mammals, including the mouse, lack melanogenically active melanocytes in their adult truncal epidermis; instead, melanin is produced in the hair follicle bulb. There is, however, considerable variation in pigment patterns within and between furred mammals; for example, perifollicular melanocytes extend to the dermis in the hairy truncal skin of the adult Syrian golden hamster (602). The highly variable color of murine pelage reflects variation in the copolymerization of eu- and pheomelanins, which results in the production of black, brown, yellow, gray, or white hair fibers. The light mutation at the brown locus results in the presentation of melanin only at the hair fiber tips due to premature death of follicular melanocytes (339). The silver mutation in mice is also associated with progressive graying caused by loss of melanocytes. Silver melanin is similar to brown and light melanins in those light-silver animals producing a diffuse "soluble" melanin within degenerating melanocytes. Studies performed on the yellow (Ay/a) mouse and on the tortoise-shell guinea pig, with its potential to develop black, red, yellow, or white hair, have shown that Lowest levels are associated with black eumelanic hair, while highest levels are found in animals with lighter colors, and largely pheomelanosomes (40). Many of these color patterns map genetically to the extension locus (see elsewhere in this review). However, hair shaft color reflects not just the quantity and quality of the pigments produced by hair bulb melanocytes, but also the manner in which they are transferred to the hair shaft. Thus mouse coat color mutations, besides being associated with differences in melanin synthesis, can also be due to abnormalities in the formation of the melanosome and their transference to keratinocytes. These mutations are detected on qualitative and quantitative electron microscopy (224). For example, the albino locus is associated with a reduction in melanosome size, but data suggest that the albino locus, in addition to involve that structure, also has a functional (tyrosinase) role in the differentiation of mouse hair-bulb melanosomes. Data on melanosome length-to-width ratios indicate that the agouti locus determines melanosome shape, either spherical or elliptical. The agouti locus, even in the absence of melanization, directs melanosome shape via synthesis and deployment of agouti-locus-encoded matrix proteins, or via other structural actiors. Abnormal transfer of melanin granules into the hair shaft can also lead to variation in pigmentation of mouse coat. An example of this is the dilute mouse, where reduction in coat color is due to mutation of the gene encoding for myosin Va (885). Thus, whereas mature melanosomes of normal melanocytes are located at the dendrite tips, in dilute mice they are retained in the perinuclear region of the cell due to a defect in protein motor capacity to transport melanosome along the cytoskeletal tracks to the dendrite tips. The discovery of this mouse gene, later identified as the first candidate gene for the Griscelli locus (Griscelli syndrome patients display severe immunodeficiency with diluted hair pigmentation), has stimulated a new field of study concerned with the function of molecular motors in vesicle/organelle transportation (849). There is no evidence for differences in melanosome biogenesis between follicular and epidermal melanocytes. Thus, in black hair follicles, melanocytes contain the largest number and most electron-dense melanosomes (eumelanosomes), each with a fibrillar matrix; in brown hair, bulb melanocytes are somewhat smaller, and in blonde hair melanosomes are poorly melanized, often with only the melanosomal matrix visible. Red hair pheomelanosomes contain a vesicular matrix, but melanin is deposited irregularly, in blotches. Of interest, both eumelanogenic and pheomelanogenic melanosomes can coexist in the same human cell (316), but not within the same pathway. These structural principles apply to follicular melanocytes and also probably to human epidermal melanocytes and rodent cutaneous melanocytes (181, 515). It was recently proposed that late endosomes may be the initial sites of stage I melanosome formation (333, 335, 456, 608); further maturation would be driven by delivery of structural and enzymatic proteins, and by their activation followed by substrate delivery. Others believe that formation of melanosomes is driven by independent, but overlapping, mechanisms (384). One suggests that the enzymatic proteins required for melanogenesis are delivered via coated vesicles to melanosomes that originate from the endoplasmic reticulum and Golgi (335). Electron microscopy of melanosome development during eumelanogenesis in normal melanocytes (af), of pheomelanogensis (gj), and of granular melanosomes in melanoma cells that synthesize eumelanin (k). Melanogenesis commences when tyrosinase and other relevant enzymes are cleaved, and initiation of activity may depend on an acidic environment provided by proton pumps (454, 601).
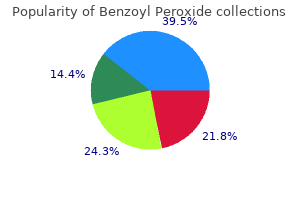
He had sensory deficits for touch and pain sensations in the distribution of the stocking area of both feet and lower legs and a mild form of facial nerve palsy involving the right side of the face acne 4 hour order benzoyl 20 gr without a prescription. The patient was suspected of having Guillain-Barrй syndrome and was admitted to the hospital for observation skin care 10 year old 20gr benzoyl fast delivery. The cause of this disease is unknown acne wash with benzoyl peroxide purchase benzoyl 20 gr with visa, although it is believed to be viral and involve the immune system acne holes in face benzoyl 20gr without a prescription. Histologically, the peripheral nerves show focal scattered areas of demyelination with an accumulation of lymphocytes and macrophages. As the myelin is lost, the axons are left naked and the Schwann cell bodies remain intact. In the majority of patients,recovery occurs in 2 to 4 weeks as remyelination occurs. Hospitalization is necessary in the early stages because the disease can spread rapidly to involve the intercostal and phrenic nerves, resulting in paralysis of the intercostal muscles and diaphragm. For the same reason, the coughing and swallowing reflexes should be watched carefully. A physician would find this disease impossible to understand without a knowledge of the structure of peripheral nerves. The process of nerve degeneration is fast and can take place in nerves in the central and peripheral nervous systems. Because so much research today is being devoted to investigating why regeneration in the central nervous system ceases within 2 weeks, the histologic changes that occur must be learned. Formation of Myelin Myelin sheaths begin to form before birth and during the first year postnatally. In the peripheral nervous system, the nerve fiber or axon first indents the side of a Schwann cell. Later,as the axon sinks farther into the Schwann cell, the external plasma membrane of the Schwann cell forms a mesaxon, which suspends the axon within the Schwann cell. Subsequently, it is thought, the Schwann cell rotates on the axon so that the plasma membrane becomes wrapped around the axon in a spiral. The direction of the spiral is clockwise in some segments and counterclockwise in others. To begin with, the wrappings are loose, but gradually the cytoplasm between the layers of the cell membrane disappears, leaving cytoplasm near the surface and in the region of the nucleus. The thickness of the myelin depends on the number of spirals of Schwann cell membrane. Some nerve fibers are surrounded by only a few turns of the membrane, while others have as many as 50 turns. In electron micrographs of cross sections of mature myelinated nerve fibers, the myelin is seen to be laminated. The lighter minor dense line, about 10 nm thick, is formed by the approximation of the outer surfaces of adjacent plasma membranes and is made up of lipid. The fused outer protein layers of the plasma membranes are very thin and form a thin intraperiod line situated in the center of the lighter lipid layer. At the node of Ranvier, two adjacent Schwann cells terminate,and the myelin sheaths become thinner by the turning off of the lamellae. The incisures of Schmidt-Lanterman are seen on longitudinal sections of myelinated nerve fibers. They represent areas where the dark major dense line is not formed as a result of the localized persistence of Schwann cell cytoplasm. This persistence of cytoplasm involves all the layers of the myelin,and thus,there is a continuous spiral of cytoplasm from the outermost region of the Schwann cell to the region of the axon. This spiral of cytoplasm may provide a pathway for the conduction of metabolites from the surface region of the Schwann cell to the axon. Bundles of nerve fibers found in the central nervous system are often referred to as nerve tracts. Two types of nerve fibers are present in the central and peripheral parts of the nervous system: myelinated and nonmyelinated. Myelinated Nerve Fibers A myelinated nerve fiber is one that is surrounded by a myelin sheath. The myelin sheath is not part of the neuron but is formed by a supporting cell. In the central nervous system, the supporting cell is called the oligodendrocyte; in the peripheral nervous system, it is called the Schwann cell. The myelin sheath is a segmented, discontinuous layer interrupted at regular intervals by the nodes of Ranvier. In the central nervous system, each oligodendrocyte may form and maintain myelin sheaths for as many as 60 nerve fibers (axons). In the peripheral nervous system,there is only one Schwann cell for each segment of one nerve fiber. Nerve Fibers 73 Epineurium Perineurium Endoneurium Schwann cells Mesaxon Schwann cell Node of Ranvier Schwann cell cytoplasm Nonmyelinated nerve fiber Myelin sheath Axon cut transversely Figure 3-2 Exploded view of a peripheral nerve showing the connective tissue sheaths and the structure of myelinated and nonmyelinated nerve fibers. Nerve Fibers 75 Axon Cytoplasm Plasma membrane Major dense line (period line) Axon Minor dense line (intraperiod line) Axolemma Schwann cell Basement membrane Nucleus A Schwann cell cytoplasm Basement membrane B Node of Ranvier C Axolemma Axon cytoplasm E Major dense line (period line) Mesaxon Minor dense line (intraperiod line) Nucleus of Schwann cell D Figure 3-4 A myelinated nerve fiber in the peripheral nervous system. E: A longitudinal section of a mature myelinated nerve fiber showing a node of Ranvier. A number of nonmyelinated axons are enclosed in the peripheral cytoplasm of a Schwann cell (top). At the node, two adjacent Schwann cells terminate, and the myelin sheaths become thinner by the turning off of the lamellae. B: Schematic diagram of a myelinated nerve fiber in which the myelin sheath has been unrolled. In the central nervous system, oligodendrocytes are responsible for the formation of the myelin sheaths. The plasma membrane of the oligodendrocyte becomes wrapped around the axon, and the number of layers will determine the thickness of the myelin sheath. The nodes of Ranvier are situated in the intervals between adjacent oligodendrocytes. A single oligodendrocyte may be connected to the myelin sheaths of as many as 60 nerve fibers. For this reason, the process of myelination in the central nervous system cannot take place by rotation of the oligodendrocyte on the axon, as did the Schwann cell in the peripheral nervous system. It is possible that myelination in the central nervous system occurs by the growth in length of the process of the oligodendrocyte, the process wrapping itself around the axon. There are incisures of Schmidt-Lanterman in nerve fibers of the central nervous system. Table 3-1 provides a summary of facts concerning myelination in the central and peripheral nervous systems. In the peripheral nervous system, each axon,which is usually less than 1 m in diameter,indents the surface of the Schwann cell so that it lies within a trough. As many as 15 or more axons may share a single Schwann cell, each lying within its own trough or sometimes sharing a trough. In some situations, the troughs are deep and the axons are embedded deep in the Schwann cells, forming a mesaxon from the Schwann cell plasma membrane. The Schwann cells lie close to one another along the length of the axons, and there are no nodes of Ranvier. In areas where there are synapses or where motor transmission occurs, the axon emerges from the trough of the Schwann cell for a short distance, thus exposing the active region of the axon. In the central nervous system, nonmyelinated nerve fibers run in small groups and are not particularly related to the oligodendrocytes. Schwann cell nucleus Axon Myelin sheath Nonmyelinated axons Schwann cell cytoplasm Processes of Schwann cells Figure 3-8 Electron micrograph of a transverse section of a myelinated nerve fiber and several nonmyelinated nerve fibers. Smooth muscle fiber Figure 3-9 Autonomic neuromuscular junction between a nonmyelinated axon and a smooth muscle fiber. Between the individual nerve fibers is a loose, delicate connective tissue referred to as the endoneurium. The connective tissue sheaths serve to support the nerve fibers and their associated blood vessels and lymph vessels. Peripheral nerve fibers can be classified according to their speed of conduction and size (Table 3-2). Each peripheral nerve consists of parallel bundles of nerve fibers, which may be efferent or afferent axons, may be myelinated or nonmyelinated, and are surrounded by connective tissue sheaths. The nerve trunk is surrounded by a dense connective tissue sheath called the epineurium.
Order 20gr benzoyl otc. Do I Have Eczema Quiz - 5 Quick Questions to Determine If You Have Eczema.